On the heels of the 1986 “Goldfish” Release Test of hydrofluoric acid (HF), Mobil engineers sought some method to prevent the HF from forming a ground-hugging toxic cloud. In his illuminating presentation at the September 22, 2018 AQMD Refinery Committee Meeting (Watch: https://youtu.be/qwo08BtEQuM?t=5108), Goldfish Test Principal Investigator Dr. Ronald Koopman stated that the affect of the HF release “was much larger than we had expected and the downwind distance was further than we had expected” and “we found that the HF that was released all flashed into an aerosol and a vapor and so there was nothing that ended up on the collection pan or in that tank — nothing was captured — and that was a great surprise to us.”
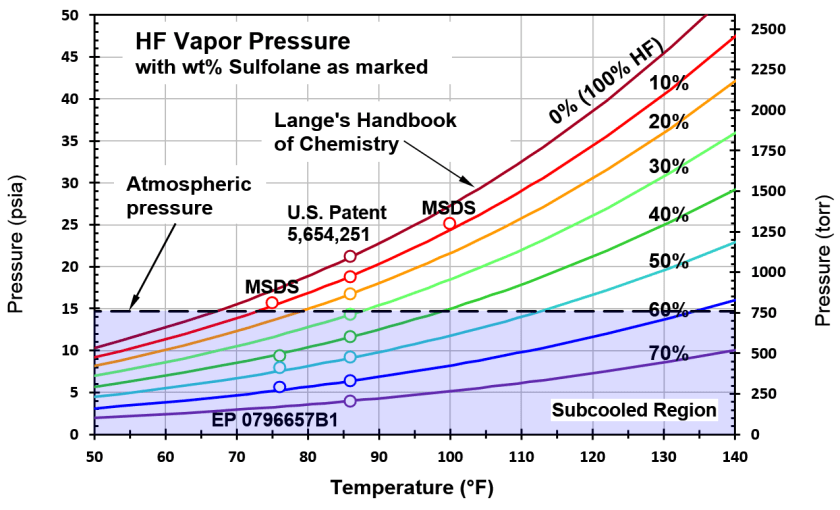
In their attempt to do something to prevent the formation of a toxic cloud, in the early 1990s, Mobil engineers settled on the additive sulfolane to suppress the vapor pressure and move HF fluid properties into the subcooled regime, where flash atomization will not occur. But, as seen in the graph above, at least 45% sulfolane by weight (45 wt%) is needed to achieve subcooled HF at a typical settler-tank temperature of 105F. Although they did not know it at the time, this level of sulfolane is far higher than the alkylation process can tolerate and still function. In refineries that use MHF, the sulfolane level is as low as 6 wt%. That’s 1 mole% or one molecule of sulfolane for every 100 molecules of HF.
–
–
Even without flash atomization, gasdynamics will break up a pressure-driven, high-velocity HF release into small droplets, and much of the droplets will evaporate into a toxic vapor before they hit the ground. To experimentally evaluate this process, in 1993 Quest Consultants were contracted by Mobil to perform a large-scale experimental evaluation in Quest’s Oklahoma facility. We learned from Quest’s Principal Investigator, John Cornwell, in his excellent presentation at the September 22, 2018 AQMD Refinery Committee Meeting (Watch: https://youtu.be/qwo08BtEQuM?t=7618) that these were the only large-scale tests conducted with MHF, and Mobil engineers selected all the test conditions. In their 1993 paper “Large-Scale Release Testing of a Modified HF Alkylation Catalyst,” Mobil engineers redacted the sulfolane concentration levels, but they are easy to figure out by comparing the test summary to U.S. Patent 528,456. The test conditions are shown by black dots in the graph below. None of the tests were with superheated MHF. Today’s 6 wt% MHF is shown on the graph below with 34oF of superheat — far more than the minimum for flash atomization into a ground-hugging cloud.
The absence of superheat in the test matrix is not the only alarming reason the experimental data is irrelevant to actual refinery operating conditions. Also, the lowest concentration of additive tested — 30 wt% — is far higher than the low level needed by the alkylation process. The first full-scale test of MHF at the Torrance refinery with 19.wt% was a failure, and the concentration had to be further reduced to the 10.wt% for the alkylation process to work. None of the additive concentrations in the 1993 Quest tests were with concentrations that would work as an effective alkylation catalyst.
Another large discrepancy between the the tests and the operating conditions at the Torrance refinery is the tank pressure. According to SCAQMD staff, the settler tanks at the Torrance refinery are mechanically pumped to a pressure of 225 psig, whereas the highest pressure selected by Mobil engineers for the tests was 140 psig. For liquid jets, higher pressure results in markedly smaller droplets, higher evaporation, and markedly smaller rainout, as the Mobil engineers acknowledged in the Summary and Conclusion of their paper: “The airborne HF reduction is improved by . . . reducing the release pressure.”
Bottom line: The rainout measurements touted from these tests are meaningless for actual refinery operating conditions. There are no large-scale tests with the low-sulfolane-level, superheated MHF used in today’s refineries to support the refineries’ false claim that MHF will not flash atomize. In fact, today’s low-sulfolane-level MHF behaves just like HF.


One thought on ““Superheated MHF Excluded from the Only Large-Scale Test Series” by Jim Eninger, Ph.D.”
Comments are closed.